 In this edition, we review (in Market Insights) the key Q1 2019 findings of the BIBA MedTech PCI Monitor—which tracks, on a quarterly basis, the percutaneous coronary intervention (PCI) procedures performed and the coronary devices used in Western Europe. We also explore the latest news (see Technology Insights) and look at pipeline developments (go to Pipeline Insights).
In this edition, we review (in Market Insights) the key Q1 2019 findings of the BIBA MedTech PCI Monitor—which tracks, on a quarterly basis, the percutaneous coronary intervention (PCI) procedures performed and the coronary devices used in Western Europe. We also explore the latest news (see Technology Insights) and look at pipeline developments (go to Pipeline Insights).
Market Insights
Key findings from Q1 2019
- Boston Scientific (with Synergy/Promus) was leader of the drug-eluting stent (DES) market, but Xience (Abbott) was the most frequently implanted DES.
- Following Absorb (Abbott) being taken off the market in September 2017, Magmaris (Biotronik) has become the most popular scaffold.
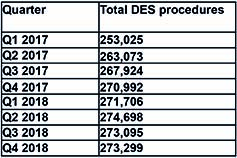
In Q1 2019, drug-eluting stents continued to be the gold standard device for performing PCI. Of 295,333 PCI procedures performed (with a permanent device) overall, 92.4% were with a DES vs. 6.5% for a bare metal stent vs. 1.1% for a bioresorbable scaffold.
The drug-eluting stent market
Source: BIBA MedTech PCI Monitor
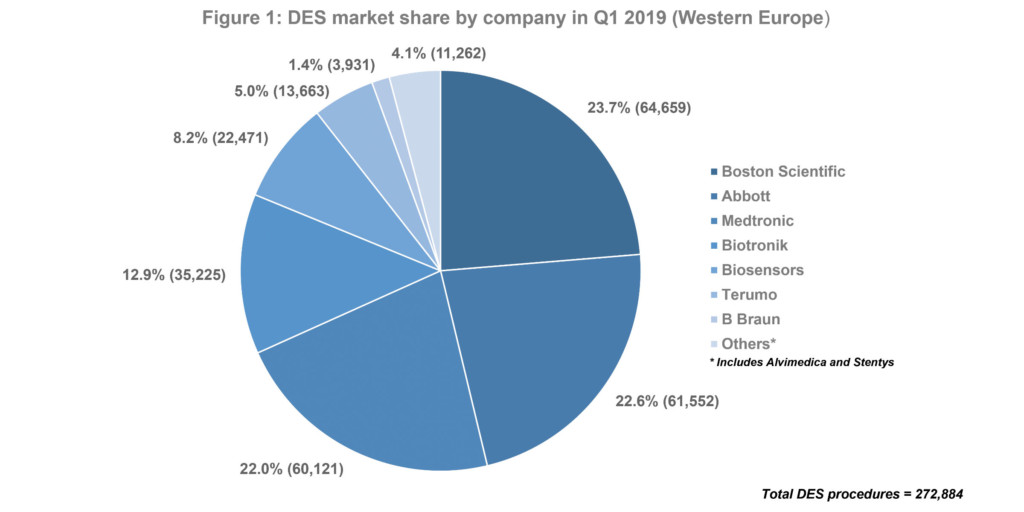
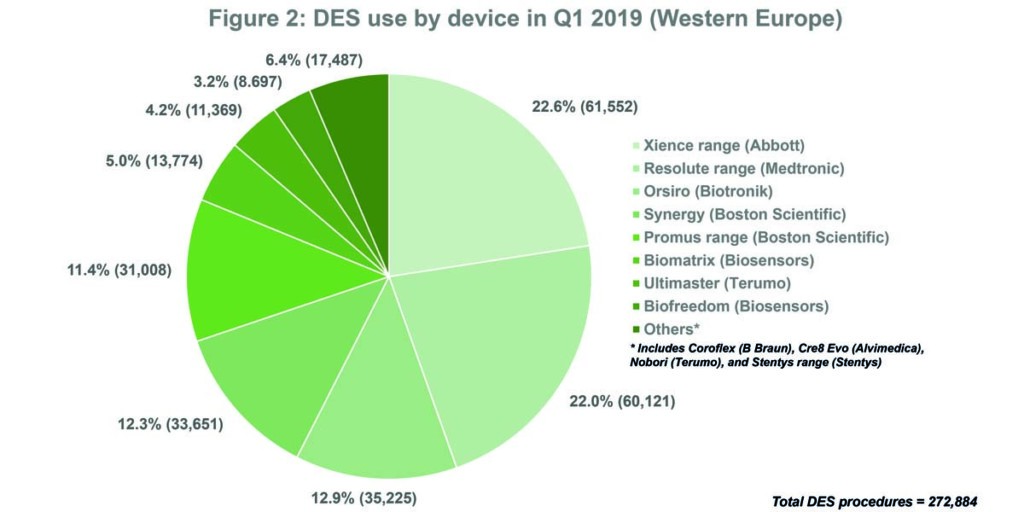
Overall, in Q1 2019, 64,659 units of a Boston Scientific device (Synergy or Promus) were implanted; this represented 23.7% of 272,884 DES implanted overall in Q1 2019—making Boston Scientific the market leader (see Figure 1). However, the most commonly used device was Xience (Abbott). See Figure 2.
Source: BIBA MedTech PCI Monitor
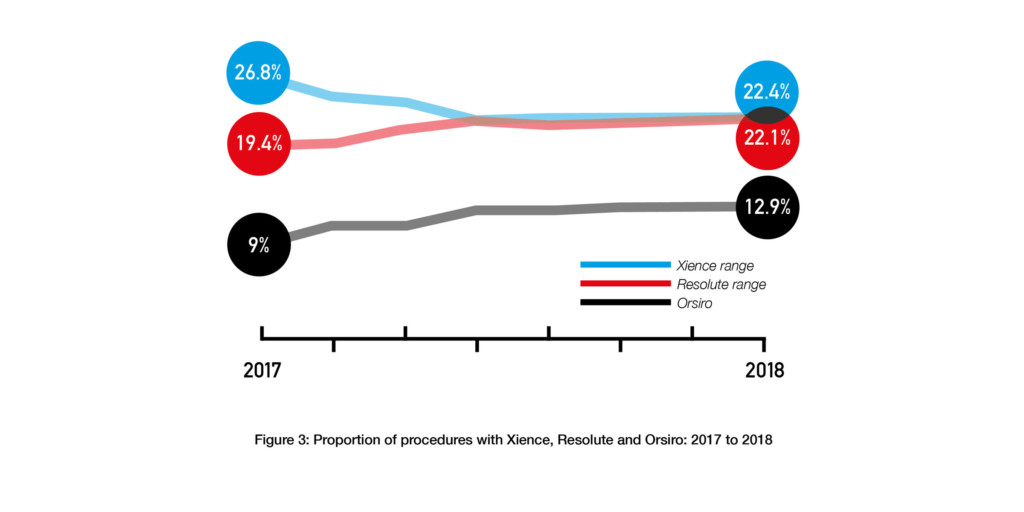
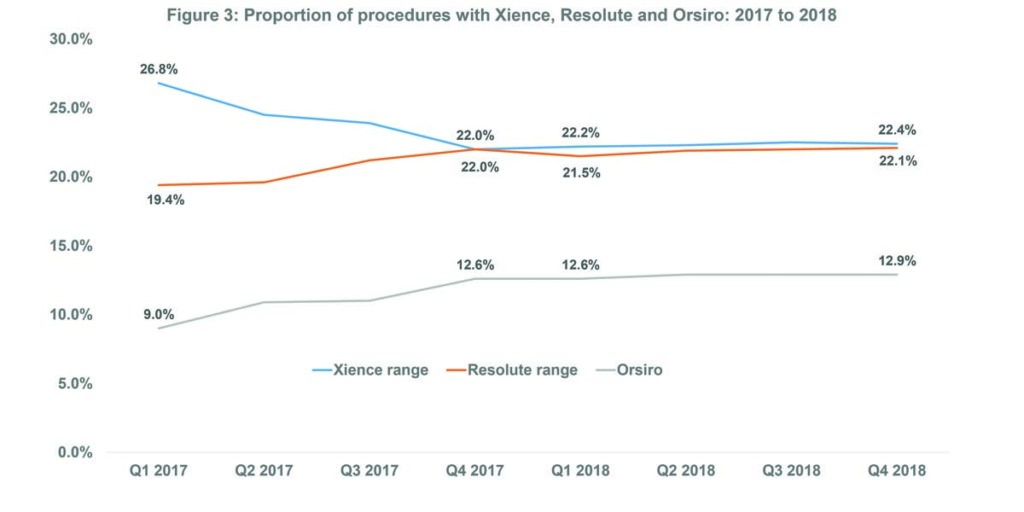
Xience has been the most commonly used DES for several years but since Q1 2017, the proportion of PCI procedures with Xience has tended to decrease. For example, 26.8% of procedures in Q1 2017 were with Xience vs. 22.4% of procedures in Q4 2018. By contrast, the proportion of procedures involving Resolute (Medtronic) has tended to increase: 19.4% in Q1 2017 vs. 22.1% in Q4 2018. The proportion of procedures with Orsiro (Biotronik) has also tended to increase over this time period. See Figure 3.
Furthermore, if these trends continue, Resolute may become the most commonly used DES by Q1 2020. Based on the available figures, approximately 280,000 PCI procedures (with a DES) are predicted to be performed in Q1 2020. Additional predictions suggest that around 23% of these procedures will be with Resolute vs. 21% with Xience.
Source: BIBA MedTech PCI Monitor
The scaffold market
In Q1 2019, Biotronik’s Magmaris device was the most commonly used bioresorbable scaffold. Of 3,173 scaffolds implanted in that quarter, 76.0% were a Magmaris device vs. 24.0% for Elixir Medical’s Desolve scaffold. Magmaris became the market leading scaffold after Abbott’s scaffold Absorb was taken off the market in Q3 2017.1 Prior to this, in Q2 2017, 61.7% of 7,432 scaffolds implanted overall were Absorb vs. 19.9% for Desolve vs. 18.4% for Magmaris.
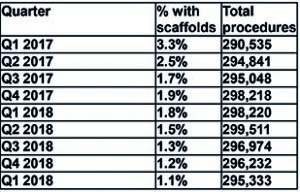
However, since Q1 2017, there has been a general decline in the use of scaffolds: 3.3% of 290,535 devices implanted overall in Q1 2017 vs. 1.1% of 295,33 devices implanted in Q1 2019. See Figure 4.
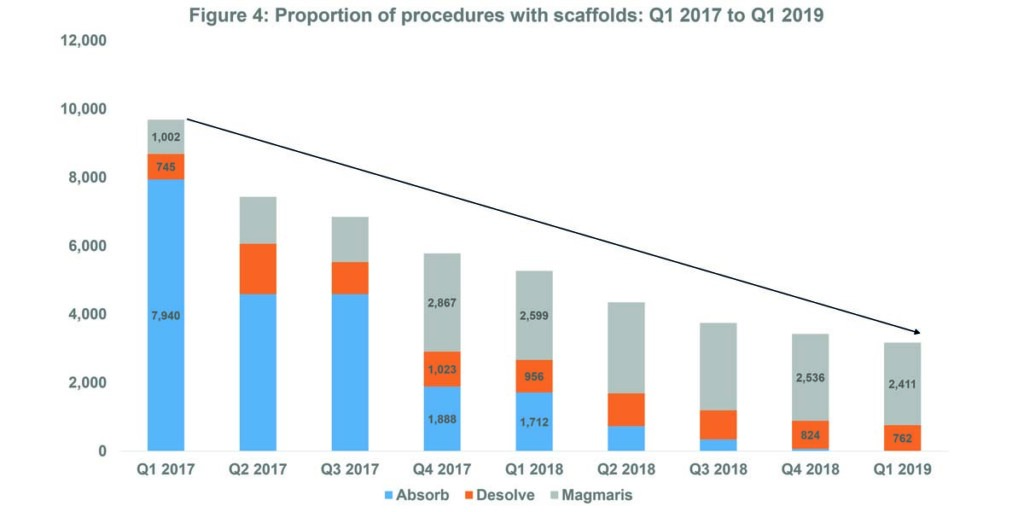
Source: BIBA MedTech PCI Monitor
Of note, data presented at the 2016 Transcatheter Cardiovascular Therapeutics (TCT) meeting (29 October–2 November, Washington, DC, USA) showed that, at three years, Absorb was associated with significantly greater late lumen loss than was Xience.2 Furthermore, two-year data from the ABSORB III study presented at the 2017 American College of Cardiology (ACC) scientific session (17–19 March, Washington, DC, USA) showed that rate of target lesion failure was significantly higher with Absorb than with Xience.3 The following month (April), Abbott wrote a letter to European physicians to say that Absorb would only be available through clinical registries at “selected sites and institutions”.4 Eventually, in September 2017, the company announced that it was taking the device off the market because of “poor commercial sales”.1
Technology Insights
Data suggest benefits of ultrathin stents may emerge in the long term

Three-year data from the BIO-RESORT trial, published in JAMA Cardiology, show that patients with small vessels who undergo percutaneous coronary intervention (PCI) with an ultrathin strut drug-eluting stent (Orsiro, Biotronik) require fewer repeat revascularisations than those who undergo PCI with a thin strut drug-eluting stent (Resolute Integrity, Medtronic). Previous findings from BIO-RESORT indicated that if ultrathin struts do provide an advantage, they emerge at two years.
The BIO-RESORT study compared three different stents: the ultrathin strut (60μm) Orsiro, which has a cobalt-chromium platform with a biodegradable polymer that elutes sirolimus; the very thin-strut (71μm) Synergy (Boston Scientific), which has a platinum-chromium platform with a biodegradable polymer that elutes everolimus; and the thin strut (91μm) Resolute Integrity, which has a cobalt-chromium platform with a durable polymer that elutes zotarolimus.5 The composite primary endpoint was target lesion failure, which was defined as cardiac death, target vessel-related myocardial infarction, or clinically-indicated target lesion revascularisation.
Writing in JAMA Cardiology, Rosaly A Buiten (Department of Cardiology, Thorax Centrum Twente, Medisch Spectrum Twente, Enschede, The Netherlands) and others say that a lower strut thickness may be particularly beneficial for patients with small target vessels because “thicker struts and a smaller in-stent lumen diameter are known to be independent predictors of restenosis in coronary stenosis”. “This can be explained by the greater relative effect of the strut size on lumen obstruction in small vessels,” they add.
Therefore, in this pre-specified analysis of BIO-RESORT, Buiten et al reviewed data for patients with small vessels. At two years, there was a significant difference in target lesion failure between patients who received Orsiro and those who received Resolute Integrity: 5.2% vs. 8.7%, respectively (p=0.03). Target lesion revascularisation was also significantly reduced with Orsiro after two years: 1.7% vs. 4.4% for Resolute Integrity (p=0.01). “Landmark analysis between one-year and three-year follow-up showed that the difference in target lesion revascularisation between sirolimus-eluting stents and zotarolimus-eluting stents emerged during the second year of follow-up (1% vs. 3.7%; p=0.006). Landmark analyses comparing everolimus-eluting stents and zotarolimus-eluting stents showed no significant difference,” the authors note.
The results of this pre-specified analysis into BIO-RESORT support previous findings that have suggested that if there are benefits of using an “ultrathin” stent over a “thin” stent, they only emerge after one year. The one-year results of BIO-RESORT did not indicate a difference between Orsiro and Resolute Integrity, but a landmark analysis reviewing data between one and two years of follow-up found that Orsiro patients had significantly lower target lesion revascularisation than Resolute Integrity patients: 0.6% vs. 1.5%; p=0.04.6,7
Furthermore, a meta-analysis comparing the ultrathin stents Orsiro, MiStent (Stentys), and BioMime (Meri) with the thicker strut stents Xience (Abbott), Resolute Integrity, and Nobori (Terumo) found that the ultrathin struts were associated with a 16% reduction in target lesion failure at one year.8 However, it did not find a difference in target lesion revascularisation between these devices. In their paper, Buiten et al observe: “The findings of our study suggest that the meta-analysis may not have been able to show a significant benefit of ultrathin stents because advantages may emerge after the one-year follow-up”.

Resolute Onyx
That the benefits of an ultrathin stent may only appear after one year may explain why the BIONYX study found that Resolute Onyx (Medtronic)—which has a strut thickness of 81μm (i.e. a thin strut stent)—was non-inferior to Orsiro for a combined safety and efficacy endpoint (target vessel failure) at one-year in all-comer patients.5 Furthermore, while the rate of definite or probable stent thrombosis was low in both groups, it was significantly lower in the Resolute Onyx group: 0.1% vs. 0.7% for Orsiro (p=0.0112). Speaking to Cardiovascular News about this trial, David Kandzari (Piedmont Heart Institute, Atlanta, USA) referenced the BIO-RESORT trial.7 Given that the trial did not start to show a difference between Resolute Integrity and Orsiro until the two-year point, he commented: “It will be interesting to see if the two-year findings of BIONYX continue to show no significant difference between Orsiro and Resolute Onyx.” He added that the stent thrombosis findings were “surprising” and were not consistent with previous studies of the Resolute range nor with “any finding for a drug-eluting stent in such a broad, unselected patient population”. “It is a very favourable result but it may be more of a chance finding,” Kandzari said.
Orsiro vs. Xience
According to Kandzari, that Orsiro was chosen as the comparator stent rather than the “gold standard” Xience stent (Abbott) was “noteworthy”. Indeed, the BIONYX investigators Clemens von Birgelen (Department of Cardiology, Thorax Centrum Twente, Enschede, The Netherlands) and others write in the Lancet that they chose Orsiro as the comparator because it has “shown excellent efficacy and safety outcomes in several randomised trials”. More importantly, they report that BIOFLOW V found that Orsiro “outperformed” Xience at one year.
In this study, at one year, target lesion failure was significantly lower with Orsiro than with Xience (which has strut thickness of 81μm): 6% vs. 10%; respectively; p=0.04. Furthermore, the BIOFLOW V investigators Kandzari et al observe: “In a pooled analysis with two additional randomised trials comparing these stents, the Bayesian posterior probability of non-inferiority for the biodegradable polymer was 100% and the posterior probability for superiority was 97%.”10 The two-year results also showed target lesion failure to be lower with Orsiro: 7.5% vs. 11.9% for Xience (p=0.015). This result, according to Kandzari et al, was driven by differences in target vessel myocardial infarction and ischaemia-driven target lesion revascularisation.10
However, of note, Bangalore et al (the authors of the aforementioned meta-analysis) comment that the Bayesian analysis of the one-year findings combined data from two earlier, smaller randomised trials and, thus, “whether newer-generation ultrathin drug-eluting stents truly improve clinical outcomes in comparison with contemporary thicker strut second-generation drug-eluting stents, and, if so, whether such results are confined to the Orsiro stent is uncertain”.8
Additionally, the BIOSCIENCE study also compared Orsiro with Xience and found that, at five years, the rate of target lesion failure was similar between groups.7 It also showed that Orsiro was associated with a significantly higher rate of all-cause mortality (driven by a higher rate of non-cardiovascular death with Orsiro). The investigators Thomas Pilgrim (Department of Cardiology, Inselspital, University of Bern, Bern, Switzerland) and colleagues write in the Lancet: “Although the observed difference in this trial might be a chance finding, it warrants further observation during long-term follow-up of ongoing studies.”11
Kandzari, in the interview with Cardiovascular News, stated that comparing data from two different trials such as BIOFLOW V and BIOSCIENCE was difficult. But, he noted that there were “very important differences in methodology between the two trials”. “For example, being a pivotal trial, BIOFLOW V had detailed endpoint definitions and ascertainment such as periprocedural biomarker assessments for periprocedural myocardial infarction—but the use of such assessments and definitions were not consistent with BIOSCIENCE,” Kandzari explained. He also echoed Pilgrim et al’s comments that the higher rate of all-cause mortality with Orsiro (in BIOSCIENCE) may be a “chance finding”, noting that “most if not all” people in the interventional cardiology community saw it as such.
Further research
Whether ultrathin-strut stents do provide benefits over thin struts needs further investigation; specifically, as Bangalore et al note, if the benefits seen with Orsiro in some of the studies is a class effect or just relate to Orsiro. Additionally, so far, strut thickness is not the only difference between Orsiro and the stents it has been compared to. For example, it has a biodegradable polymer whereas both the Xience range of stents and the Resolute range of stents have permanent polymers. Also, Orsiro elutes sirolimus whereas Xience and Resolute elute, respectively, everolimus and zotarolimus. Concluding their paper on the BIO-RESORT pre-specified analysis, Buiten et al say: “Further research is required to definitely answer the question of whether the difference in stent strut thickness is the main driver of observed between-stent difference.”
Top findings
Meetings
- EuroPCR 2019 (21–24 May, Paris, France): The two-year results from the MeRes-1 trial indicate that the second-generation bioresorbable scaffold (MeRes 100, Meril) is associated with a high rate of acute success, a very low rate of major adverse clinical events (MACE) of 1.87%, and no scaffold thrombosis.12
- EuroPCR 2019: Post-PCI microcatheter-based fractional flow reserve (FFR) is feasible and a FFR value of <0.90 is common. Furthermore, a suboptimal post FFR value of <0.90 may be associated with more stent thrombosis and myocardial infarction at follow-up.13
- EuroPCR 2019: Optical coherence tomography (OCT)-based FFR is a novel and fast method allowing assessment of flow-limiting coronary stenosis without pressure wire and induced hyperaemia. The good diagnostic accuracy and low observer variability bear the potential of improved integration of intracoronary imaging and physiological assessment.14
- EuroPCR 2019: Guidance with 3D-optical coherence domain imaging was superior to guidance with angiography in acute incomplete strut apposition for bifurcation PCI.15
- EuroPCR 2019: Short duration (one month) of triple therapy with a P2Y12 inhibitor was not superior to six-month duration triple therapy with a P2Y12 inhibitor in terms of less bleeding events in patients with atrial fibrillation who have undergone PCI.16
- EuroPCR 2019: Interim results from a prospective, worldwide registry of the Ultimaster stent (Terumo) that had >50% of patients with acute coronary syndromes indicate that the device was associated with remarkable safety and efficacy. Specifically, the rate of target lesion failure was 5% and the rate of definite/probably stent thrombosis was 1%.17
- EuroPCR 2019: The BioMime Morph (Meril) appears to be safe and effective in very long (up to 56mm) coronary lesions in native coronary vessels with a reference diameter of 2.25–3.5mm. The long-tapered drug-eluting stent may be an “extremely interesting alternative” for long diffused de novo coronary lesions with tapered anatomy in routine clinical practice.18
- EuroPCR 2019: Ticagrelor monotherapy after one-month dual antiplatelet therapy (DAPT), compared with 12-month DAPT, provides a significant net clinical benefit (net adverse clinical events) for patients who have undergone complex PCI but not those who have not undergone complex PCI. Therefore, ticagrelor monotherapy (after one-month DAPT) may be a better alternative to the standard DAPT in patients with complex PCI.19
Journals
- Annals of Internal Medicine: A pilot study has found that patients who received comics or “medical graphic narratives”, as well as standard informed consent protocols (official consent form and a conversation with a physician), had greater comprehension of the coronary angiography procedure that they underwent and less anxiety than those who only received the standard protocols.20
- Journal of the American College of Cardiology (JACC): Nearly two thirds (64.3%) of cath lab staff believe that the financial/administrative burdens of meeting PCI public reporting requirements are not outweighed by the potential benefits in terms of improving quality of care.21
- JACC: Between 2000 and 2014, in the USA, the percentage of patients with acute myocardial infarction cardiogenic shock (AMI-CS) who developed multiple organ failure increased from 15.7% to 45.5%. Furthermore, while use of angiography and PCI in AMI-CS patients has increased over this time period, the use of these procedures in patients with AMI-CS and multiple (or single) organ failure has remained low.22
- JACC: An implantable cardiac system could detect early ST-segment deviation and alert patients about a potential occlusive event. Although the trial did not meet its prespecified primary efficacy endpoint (the composite primary efficacy endpoint was cardiac/unexplained death, new Q-wave myocardial infarction, or detection to presentation time more than two hours), results suggest that the device may be beneficial among high-risk patients in potentially identifying asymptomatic events.23
- JACC: Cardiovascular Interventions: Although >80% of stable patients with ST-segment elevation myocardial infarction (STEMI) are treated in the ICU after primary PCI, the risk for developing a complication requiring ICU care is 16%. Implementing a risk-based triage strategy, inclusive of factors such as degree of reperfusion delay, could optimise ICU use for patients with STEMI.24
Pipeline Insights

- May 2019: Medtronic launched Telescope guidance catheter onto the global market. A press release reports that this launch marks the company’s “entrance into the guide extension catheter market”. It adds that the guide extension catheter combines a solid, round pushwire with a coil-reinforced hydrophilic-coated distal segment. Furthermore, Telescope also features a flexible TruFlex soft polymer tip, which is designed to responsively bend and deflect during use.1
- May 2019: According to results from the BOWIE (Benefits of obtaining information for planning with non-invasive FFRCT prior to invasive evaluation) study, use of the investigational HeartFlow Planner (HeartFlow) led to a change in treatment strategy in 45% of patients with coronary artery disease and reduced the need for invasive physiology. Eric van Belle (head of the Lille Heart & Lung Institute, Lille, France), principal investigator for BOWIE, presented the study at the 2019 EuroPCR meeting (21–24 May, Paris, France).2
- May 2019: Shockwave Medical has appointed Keith D Dawkins as its chief medical officer. Prior to working for Shockwave Medical, Dawkins was the global chief medical officer and executive vice president to Boston Scientific.3
- May 2019: The interventional cardiologists Michael Haude, Faizel Lorgat, and Holger Nef have joined the medical board of Robocath—which has developed a robotic-assisted system for coronary angioplasty (R-One, which received the CE mark in February 2019). The interventional cardiologists Alberto Cremonesi, Alain Cribier, Jean Fajadet, Eric Durand, Rémi Sabatier, and Gregg Stone are already on the board.4
- April 2019: PK Papyrus covered coronary stent (Biotronik) was launched onto the US market. A press release reports that this is the first FDA-approved device for acute coronary perforation in nearly 20 years.5
- February 2019: The US FDA approved the Biotronik’s Orsiro stent. The stent has a biodegradable polymer, elutes sirolimus, and has an “ultrathin” strut platform. According to a press release, the stent is the “first and only” ultrathin drug-eluting stent to outperform the clinical standard, Xience. Orsiro received CE marking in 2011 and has been used to treat more than one million patients worldwide to date.6
- February 2019: The Resolute range (Medtronic), including Resolute Integrity and Resolute Onyx, received US FDA approval for the treatment of patients with de novo chronic total occlusion.7
- February 2019: After receiving the CE mark for the device, iVascular launched its Navitian coronary microcatheter onto the market. A press release reports that the microcatheter can be used to facilitate, guide and support a guidewire while accessing the coronary system, the exchange of guidewire, and the injection of radiopaque contrast media or saline solutions.8
References
- BIBA Medical staff. “Low commercial sales” prompt Abbott to pull the plug on selling Absorb. Cardiovascular News 2017.
- BIBA Medical staff. Three-year data fail to show Absorb to be superior or even non-inferior to Xience. Cardiovascular News 2016.
- BIBA Medical staff. Two-year rate of target lesion failure significantly higher with Absorb. Cardiovascular News 2017.
- BIBA Medical staff. Absorb GTS to be restricted to registry use only in Europe. Cardiovascular News 2017.
- Buiten RA, Ploumen EH, Zocca P, et al. Outcomes in patients treated with thin-strut, very thin-strut, or ultrathin-strut drug-eluting stents in small coronary vessels: A prespecified analysis of the randomized BIO-RESORT trial. JAMA Cardiol Epub.
- BIBA Medical staff. Orsiro may be the new benchmark for comparison in drug-eluting stent trials. Cardiovascular News https://bit.ly/2S3QZa1 (date accessed 5 July 2019).
- Kok MM, Zocca P, Buiten RA, et al. Two-year clinical outcome of all-comers treated with three highly dissimilar contemporary coronary drug-eluting stents in the randomised BIO-RESORT trial. EuroIntervention 2018; 14 (8) :915–23.
- Bangalore S, Toklu B, Patel N, et al. Newer-generation ultrathin strut drug-eluting stents versus older second-generation thicker strut drug-eluting stents for coronary artery disease. Circulation 2018; 138(20): 2216–26.
- von Birgelen C, Zocca P, Buiten RA, et al. Thin composite wire strut, durable polymer-coated (Resolute Onyx) versus ultrathin cobalt-chromium strut, bioresorbable polymer-coated (Orsiro) drug-eluting stents in allcomers with coronary artery disease (BIONYX): an international, single-blind, randomised non-inferiority trial. Lancet 2018; 392 (10154): 1235–45.
- Kandzari DE, Koolen JJ, Doros G, et al. Ultrathin bioresorbable polymer sirolimus-eluting stents vs thin durable polymer everolimus-eluting stents. J Am Coll Cardiol 2018; 72 (25): 3287–97.
- Pilgrim T, Piccolo R, Heg D, et al. Ultrathin-strut, biodegradable-polymer, sirolimus-eluting stents versus thin-strut, durable-polymer, everolimus-eluting stents for percutaneous coronary revascularisation: 5-year outcomes of the BIOSCIENCE randomised trial. Lancet 2018; 392(10149): 737–46.
- Abizaid A. Presentation at EuroPCR 2019.
- Diletti R. Presentation at EuroPCR 2019.
- Tu S. Presentation at EuroPCR 2019.
- Onuma Y. Presentation at EuroPCR 2019.
- Hosi T. Presentation at EuroPCR 2019.
- Roffi M. Presentation at EuroPCR 2019.
- Agostoni P. Presentation at EuroPCR 2019.
- Serruys PW. Presentation at EuroPCR 2019.
- BIBA Medical staff. Comics may help patients to better understand coronary interventions. Cardiovascular News 2019.
- BIBA Medical staff. Majority of cath labs feel burdens of PCI public reports outweigh benefits. Cardiovascular News 2019.
- BIBA Medical staff. Prevalence of multiple organ failure among cardiogenic shock patients has steadily increased. Cardiovascular News 2019
- Gibson CM, Holmes D, Mikdadi G, et al. Implantable cardiac alert system for early recognition of ST-segment elevation myocardial infarction. J Am Coll Cardiol 2019; 73(15): 1919–92.
- Shavadia JS, Chen AY, Fanaroff AC, et al. Intensive care utilization in stable patients with ST-segment elevation myocardial infarction treated with rapid reperfusion. JACC Cardiovasc Interv 2019; 12(8): 709–17.
- Medtronic launches Telescope guide extension catheter to support complex coronary cases.
- Use of HeartFlow Planner leads to change in treatment strategy in nearly half of patients with coronary heart disease.
- BIBA Medical staff. Keith Dawkins appointed as chief medical officer at Shockwave Medical. Cardiovascular News 2019.
- Robocath strengthens its medical advisory board with international interventional cardiology experts.
- Biotronik Launches PK Papyrus covered coronary stent in the US.
- FDA approves Biotronik’s ultrathin Orsiro stent for the treatment of coronary artery disease.
- Medtronic Resolute drug-eluting Stent platform receives expanded indication for treatment of chronic total occlusion.
- BIBA Medical staff. iVascular receives CE mark for Navitian microcatheter.
For more information about BIBA Briefings Insights reports, contact Dawn Powell: [email protected]





