
Tom Oxley
Synchron recently announced the first successful implant of its minimally-invasive neural interface technology (Stentrode) as part of a trial evaluating the safety and efficacy of the technology for restoring communication in people with severe paralysis. The technology uses modular training software (BrainOs) to control assistive technologies through thought. The company’s CEO Thomas Oxley (Synchron, Campbell,) spoke to BIBA Briefings about the Synchron brain-interface system, describing it as a “bypass” for the nervous system.
Why is there a need for a device, such as Stentrode, to restore communication in patients with severe paralysis?
It is very clear from patients with severe paralysis who are not able to communicate that the lack of communication is far and away the most important [negative] element in terms of quality of life. However, currently, we do not have a technology that truly overcomes spinal or brainstem injury. Stephen Hawkins, for example, had to use a small muscle in his cheek to activate a single switch to communicate. With this technology, he wrote A Grand Design at about a rate of one word per minute.
If you took the concept of using a switch to communicate to the extreme by having multiple switches, with no limitations on the number of “switches”, then you could have a very efficient form of communication. People with disabilities as severe as Hawkins, or even milder, could have a similar level of communication as a person without any communication disabilities.
How does the Stentrode, in conjunction with the Synchron Brain-Computer interface, seek to restore communication?
We are creating a “technology bypass” that wirelessly connects the motor cortex in the brain to the brainOS app platform. The aim with the platform is to overcome the three primary features of paralysis—speech deficit, upper limb failure, and lower limb failure—and the first application is speech.
To do that, we are placing an interventional stent electrode—which we are calling “Stentrode”—into the command centre of the brain; the motor cortex. We then train patients to activate the Stentrode by “thinking” a specific set of thoughts.
How does your system differ from other neural interface devices, such as Elon Musk’s Neuralink and BrainGate, in development?
It is less invasive. The main difference is that the other technologies out there require a craniotomy; so, part of the skull needs to be removed to get to the brain. Our system, however, uses neurointerventional techniques to implant the system through the blood vessels. Though, unlike traditional neurointerventional techniques, we are targeting the venous system rather than the arterial system.
The system wirelessly sends the information from the stent to the brainOs platform. How are you ensuring the cybersecurity of the system?
The security of the system is a major area of concern that we are taking very seriously. There are lessons that we can learn from other companies that remotely access information from devices implanted into the brain (such as cochlear implants). It is not a novel problem.
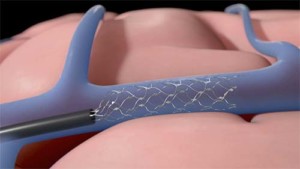
Stentrode
What data are already available for the system?
Twenty-five scientific studies on the system have been published in international peer-reviewed journals. We have looked at the risks of stenosis, thrombosis, endothelialisation, and the signal quality over time [demonstrating the long-term safety of the system]. The largest study was published in Nature Biotechnology in 2016 and was an extensive work; it took us years to put that data together.
What are the aims of the ongoing feasibility trial?
A critical part of the study will be to characterise which patients could benefit from the technology, but the primary endpoint is the safety of the system. In terms of safety, we will be monitoring the patient to determine the potential risks, including haemorrhage and thrombosis, with the system. The secondary endpoints will be the quality and the stability of the signal over time.
With the data from the study, we will begin to adjust our artificial intelligence algorithms as we learn how we can best clarify the signal that comes of the stent and use it to control the apps.
What is the inclusion and exclusion criteria of the study?
In Australia, we have approval for five patients with a range of conditions: stroke-related paralysis, motor neurone disease, spinal cord injury, and muscular dystrophy. All patients must have a positive functional MRI, to ensure the motor cortex is intact and functioning. Additionally, a neuroradiologist has to assess the vascular system to ensure that it is appropriately structured for the implant into the motor cortex. Patients do need to have a life expectancy that is greater than 12 months.
You are already in discussions with the US Food and Drug Administration (FDA) about market approval?
Yes, our broad goal is to get FDA approval; therefore, we have been in discussions with them for a couple of years. We have designed an extensive trial programme that is seeking to demonstrate the safety and efficacy of system in the way that the FDA requires for approval.
The FDA actually published, in February 2019, a guidance document (Implanted brain-computer interface devices for patients with paralysis or amputation) about getting approval because of the number of brain-interface technologies in the pipeline. They have communicated clearly what is needed to demonstrate safety, so we are working through that very long and extensive checklist.
It is too early to say when we would have market approval. It will be a number of years.





